Abstract
Ever since Harrit e.al.'s paper "Active Thermitic Material discovered" (ATM, [1]) was published in April 2009, the world of 9/11 debaters (a small world, by the way) was split into two camps:
9/11 Truthers who believe all the chips are super-secret high-tech military-grade beast of extremely energetic nano-thermite. Note the stress on „all the chips“
Skeptics who see that the chips are not all the same, are not thermitic, but very probably different kinds of paint instead.
In this post I will show that one particular chip in ATM, the one they soaked in MEK and present in Fig. 12-18, cannot possibly the same kind of material as the four chips they present in Fig. 2-11. Assuming that both represent the same material is preposterous. The most benign explanation for why the authors make that assumption is wishful thinking. We can rule out simple error or that they overlooked something, because it has been pointed out to them more than once in the past that the chips are different. A less benign, but perhaps more probable explanation would be outright fraud.
Visual comparison
Here are the chips I am talking about – first, the four chips they first present. I usually refer to them as chips (a)-(d):
As you can see, the red layers all look pretty much like they could be the same stuff, perhaps paint. Color is very similar, finish is very similar. Same goes for the gray layer, which could be metallic for all we know (and yes, Harrit e.al. figure out correctly that the gray layer is a bulk of oxidized iron). Notice that we can see and roughly measure the thickness of the red layer in the inset of Fig 2(d): It is roundabout 15µm thick.
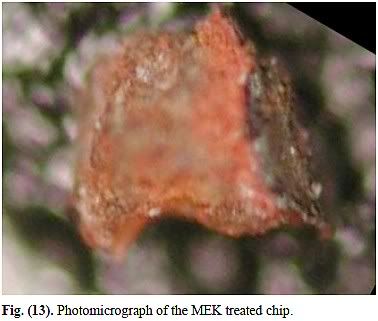
Next up, the MEK-chip:
Whoa – what's up there?? The photo is totally out of focus! So yeah it is generally some kind of red and there seems to be some gray on the right, but does it have the same finish as (a)-(d)? Frankly, I can't tell! How thick is the layer? We can't tell from the photo, but Harrit e.al. included another image. In the following, the chip is shown after they had soaked it in a solvent called MEK for 55 hours.
They explain on page 17:
The red layer of the chip was found, by visual inspection, to have swelled out from the gray layer by a factor of roughly 5 times its original thickness.
In Fig 12(b), the red layer, on the left, is between 250 and 300 µm thick, aproximately, so before soaking it was 50-60 µm. Quite a bit more than the 15 µm of the red layer of chip (d) above, eh? (In Fig. 5, it is possible to roughly measure the thickness of the red layers of chips (a) and (b): approx. 32 and 13 µm respectively). So does that MEK chip look the same as the others? Hmmmm maybe, maybe not. Maybe not.
Harrit e.al. show high-magnification BSE (a form of electron microscopy) images for chips (a)-(d) in Fig. 4, Fig. 5 and Fig 8, where you can see the grains (identified by Harrit as hematite) and platelets (almost certainly kaolin, a natural clay) in the organic matrix. Unfortunately, no such BSE images exist for the MEK-chip, so we can't compare it to chips (a)-(d). The only other data we have is XEDS.
XEDS spectra
An introduction to XEDS (SEM-EDS)
(You may skip this section if you are not interested in technical details of this method)
XEDS (X-ray energy dispersive spectroscopy, also abbreviated SEM-EDX, see Wikipedia [2]) is
...an analytical technique used for the elemental analysis or chemical characterization of a sample. […] a high-energy beam of charged particles such as electrons or protons (see PIXE), or a beam of X-rays, is focused into the sample being studied. […] The number and energy of the X-rays emitted from a specimen can be measured by an energy-dispersive spectrometer. As the energy of the X-rays are characteristic of the difference in energy between the two shells, and of the atomic structure of the element from which they were emitted, this allows the elemental composition of the specimen to be measured.
In an XEDS graph, the location of a peak along the x-axis identifies a chemical element, while the height of the peak along the y-axis is indicative of the relative abundance of the element in the sample. Please note first that equal peak heights of two different elements do not automatically mean same abundance, although this is roughly true for many elements (for example, it is true for aluminium and silicone), but not all (for example, if strontium and chromium were equally abundant by mass, then the first peak of strontium at 1.81 keV would be only about 75% the height of the chromium peak at 5.41 keV. This is also dependent on factors like “accelerating voltage and/or contaminating surface films” [3]). A second note: The lightest elements, from hydrogen (1H) typically to beryllium (4Be) don't show up at all in an XEDS graph (depending on the device, not even up to carbon or nitrogen). The next lighter elements up to chlorine (17Cl) only have one peak associated to them. Starting with potassium (18K), more than one peak may show up, but in most cases, only one or two are dominant. Last note: Peak height scales with abundance. So if you double the abundance of, say, silicone in your sample, the Si-peak will be twice as high (roughly). If you want to look up only which elements have peaks at which energy levels (measured in keV – kilo Electronvolts), you may refer to [4]. Just klick on the element symbol you are interested in, and look in the column “Edge Energies”. Usually, the K-alpha level is your first major peak, K and K-beta for secondary peaks. Elements heavier than arsenic (33As) don't have important K-levels below 10 keV and are more usually identified by L-alpha or L-beta.
The spectra of Harrit e.al.
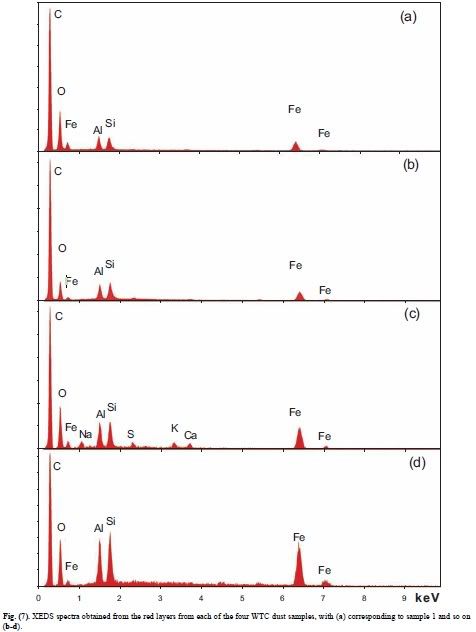
Harrit e.al. provide XEDS spectra for chips (a)-(d) in Fig. 7, and a spectrum for the MEK-chip (before soaking) in Fig 14. Let us first take a close look at all the peaks in Fig. 7 (shown below) and see if these four graphs are similar enough so we can be confident that all four show the same material. All have major peaks for 5 chemical element (from left to right, the major peaks: Carbon (C), oxygen (O), aluminium (Al), silicone (Si), iron (Fe)). In figure 7(c), Harrit e.al. have also labelled small peaks of natrium (Na), sulfur (S), potassium (K) and calcium (Ca). In addition, we think there are tiny but discernable signals for S and Ca in (a) and (b) as well, for chromium (Cr, K-alpha = 5.41 keV) in (a), (b) and (d), and titanium (Ti, K-alpha = 4.51 keV) in (d). While the small peaks could always be some sort of contamination (either on the surface, or of the minerals contained in the chips; for example, kaolin usually has small inclusions of Ti and Ca), the major elements do show up in comparable relative peak heights:
In all four chips, C is by far the highest peak, being several times (2.85x to 7.45 time, average 4.3 times) as high as the second highest, peak, O
O is the second highest in 3 graphs, and barely beaten by Si in 1. O has between 85% and 300% the peak hight of Si (average: 161%)
Si and Al follow in third and fourth place, at almost the same hight. Al has between 87% and 110% the peak hight of Si (average: 96%). This result is consistent with both elements appearing in equal molar amounts.
Fe (K-alpha) is always the fifth-highest peak, reaching between 51% and 81% of Si (average 70.5%)
Note that none contain zinc (Zn) or magnesium (Mg), and all have at most traces of Ca and S
Here is Fig. 7:
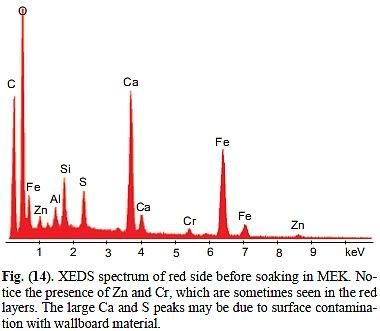
Now compare this to the MEK-chip, Fig 14:
It is very obvious that none of the characteristica of Fig. 7 are found here: For starters, Al is not among the 5 or 6 highest peaks, it is only number 7. Instead, Ca clocks in as the sceond highest peak. So let's go through the list item by item:
C is not the highest peak, it is only the 3rd-highest. Instead of being at least 2.8 times as high as O, it has only about 60% of the height of O.
O is much too abundant – relative to C (and, coincideltally, to Al) by a factor of at least 4.7
Si and Al are not about equally abundant. Si-peak is too high relative to Al by a factor of 1.8
There is way too much Fe relative to both Al and Si: Fe should be near 68% of Al, but it is actually 2.75 times as high. This means, too abundant by a factor of 4.
The Ca peak is HUGE, it should only be a trace. The S-peak is BIG, it should at most be a trace. There should be no Zn at all. There is a peak between Zn and Al that Harrit e.al. did not label, but which certainly represents Mg. There should be no signal for Mg.
Discussion
How do Harrit e.al. explain these differences between Fig 7 and Fig 14? Here's how (page 17):
The resulting spectrum, shown in Fig. (14), produced the expected peaks for Fe, Si, Al, O, and C. Other peaks included calcium, sulfur, zinc, chromium and potassium. The occurrence of these elements could be attributed to surface contamination due to the fact that the analysis was performed on the as-collected surface of the red layer. The large Ca and S peaks may be due to contamination with gypsum from the pulverized wallboard material in the buildings.
So pretty much all of the Ca, all of the S, 75% of the Fe, 80% of the O, 45% of the Si, all of the Zn, all of the Mg is contamination? Gypsum, eh?
Here are three XEDS graphs for gypsum from the WTC [5]: Gypsum-01, Gypsum-02, Gypsum-03
Note that in the first two of the graphs, S peak is higher (by about 35% and 32%) than Ca, and in the third, which also has (calcium-?) carbonate, S is 33% lower than Ca. McCrone ([3] page 638) has S about 9% lower. This is to be expected, as the chemical formula for gypsum is CaSO4·2H2O - notice how Ca and S both have one atom in that molecule, their molar abundance is equal, their atomic weight is not much different (S: 32; Ca: 40; that's a ratio of 1:1.25). So, if you assume that gypsum is a major contaminant in Fig. 14, you must take off as much (+/- 33%) S as Ca – until you run out of S. Now, in Fig 14, Ca is 3 times as high as S. If you claim all of the S is from gypsum, and you remove all of it, and If I grant you that you may remove 33% more Ca than S, then the Ca peak is still almost as high as Fe, and higher than Si and Al. And the Fe-peak is still too high relative to C and Al, Si is too high relative to Al. So obviously, even if gypsum explaines all of the “contamination” with S, it would still constitute only a minor part of all of the “contamination”. In fact, to make Fig. 14 look similar to Fig. 7, you must
remove 80% of the oxygen (highest peak)
remove 95% of the calcium (2nd highest peak)
remove 75% of the iron(4th highest peak)
remove 45% of the silicone (5th-highest peak)
remove 95% of the sulfur (6th-highest peak)
remove all of the Zn
remove all of the Mg
remove most of the Cr
In other words: On average you must declare two thirds (65%) of the six most abundant elements to be contamination.
This is absurd. Preposterous. Wishful thinking. If not fraudulent.
Conclusion
A much better explanation is in order: Since no data exists, other than the base color and magnetic attraction, that shows that the MEK-chip is the same material as chips (a)-(d), since the visual appearance is doubtful, since the layer is too thick, and since the XEDS data shows that at least 65% of the mass of this chip is different from chips (a)-(d), the best and obvious conclusion is:
The MEK-chip is of a different material than chips (a)-(d). The assumption that the differences can be explained as contamination does not survice scrutiny and must be firmly rejected.
References
[1] Niels H. Harrit, Jeffrey Farrer, Steven E. Jones, Kevin R. Ryan, Frank M. Legge, Daniel Farnsworth, Gregg Roberts, James R. Gourley and Bradley R. Larsen: Active Thermitic Material Discovered in Dust from the 9/11 World Trade Center Catastrophe. The Open Chemical Physics Journal, 2009, 2, 7-31
[2] Energy-dispersive X-ray spectroscopy. Wikipedia, retrieved 2012/03/14
[3] Walter C. McCrone and John Gustav Delly: The Particle Atlas Edition Two, Volume III: The Electron Microscopy Atlas. Ann Arbor Science Publishers Inc., 1973, page 579
[4] Illinois Institute of Technology: Peridiodic Table. Last retrieved 2012/03/14
[5] US Geological Service: Particle Atlas of World Trade Center Dust. Open-File Report 2005–1165: On-line Report, öast retrieved 2012/03/14


Who will have the patience to argue with a troll like you?
ReplyDeleteEven Milette himself agrees with Harrit that the different graphs are due to surface contamination.
And he has ruled out the primer paint just like Harrit...because the chemical composition is completely different...
which should have been obvious to everyone from the start since the chips do not dissolve in a paint solvent, even after 55 hours, which is quite a bit for chips on the milligram scale.
Yet we have morons who claim that the chips must be the primer paint.
PS. the chips explode and outperform TNT when ignited at 430 degrees(C), but the paint is stable at over 800, and obviously does not explode.
Get a clue!
Hello Anonymous,
Deletethanks for your comment! I'll courteously reply with a few questions and remarks to each point:
1. Can you tell me, with a quote perhaps, that "even Milette himself agrees with Harrit that the different graphs are due to surface contamination"? I am not talking about small blips for Na or Mg or S that frequently show up all over the place. I am talking about 80% of the highest peak, 95% of the third highest peak, and two thirds of the entire spectra.
2. In case you missed it, there was not just one primer, there were at least 2: See http://oystein-debate.blogspot.de/2012/03/another-primer-at-wtc-laclede-standard.html and Millette only ruled out one primer for several of his chips (Tnemec). He didn't even look at LaClede primer, so he didn't rule it out.
Could you please acknowledge that you now know there were at least two primers, and that, if one doesn't match, the other could still match? Or any third, fourth, fifth primer that's yet unknown?
Millette is pretty clear in his description of the red layer as PAINT, he just hasn't figured out the paint PRODUCT yet, if you read his prelim report closely.
3. Millette's chip also didn't dissolve in MEK. That's why he had to low-temp ash some ships to free the pigments. You probably don't know this, but epoxies, with their intense crosslinking, don't simply dissolve, they just become soft.
4. Millette is one such "moron". He followed accepted professional protocol for analysis of paint, and found all the ingredients of paint. But no aluminium, hence no thermite.
5. Yes. Outperforms TNT in the energy density department. You could chop off your earlobe and test in the DSC, and you will find that it, too, outperforms TNT, or thermite, in that department.
Again, you miss the point that there is at least one other primer paint, and you don't know how that performs up to 800°C, so hold your judgement.
By the way, I am working on a blog post to explain why Farrer's DSC testing doesn't prove what you think it proves.
Sure, Jesus Christ...since it happens that the main primer BS has been proven false, you are sure there must be another one that fits your theory, if you keep searching long enough..
ReplyDeleteand sure,someone must have decided to make paint that explodes at 400 degrees for the floors...it just makes sense.
And no, if you burn organic materials the power density may be higher, but the RATE of energy release will not match.
Theres a little graph for that in Harrits paper. Shit the details!
And the tremec paint DOES disolve as Harrit notes in the paper or in one of the email responses. The chips swell but remain intact.
And what happens to the tremec paint when it is ignited buddy? - it turns into goo...it does not explode, does not produce iron spheres
get a clue buddy
Hello Anonymous,
Deletethanks for you interest!
1. Tnemec maybe wasn't even the main primer, and Laclede primer is is not theory, it's fact. There was more painted surface to the LaClede floor trusses, with LaClede shop primer, than to the perimeter columns and their Tnemec primer, so perhaps LaClede was the main primer. At any rate, Harrit e.al. never considered or even acknowldegd it. See my blog post http://oystein-debate.blogspot.de/2012/03/another-primer-at-wtc-laclede-standard.html
2. Can you provide me with information what the rate of energy release actually was, and compare it to, say, the rate at which paint flakes with organic binder would burn? That would be terrific!
However I can tell you that nothing in Harrit e.al.'s tests occurred that can be correctly described as an exlosion, and indeed, the string "explosi" appears quite often in their paper to describe speculations as well as actual superthermites described in literature, such a word is never used to describe their observations of the red-gray chips. Instead, they consistently describe the reaction as "burning". So what makes you think anything exploded there? Nothing did.
3. Can you please cite a source for your claim that Tnemec does dissolve? It is not mentioned in the paper, and indeed I know already that the paints they mention in the paper, though unnamed, were not Tnemec. For details, see http://oystein-debate.blogspot.de/2010/11/origin-of-steven-jones-paint-controll.html
4. The MEK-soaked chip, which we identified as Tnemec, was not subjected to any thermal test - wasn't put in the DSC and wasn't flame-tested. So how do you know that chips would not have "turned into goo"? How do you know that Tnemec, when burned, turns into goo and doesn't produce iron spheres? Do you have a citation for that?
5. Please give me a clue - provide evidence for your claims! :)
And how come none of you listened to independent confirmations by the French dude and then Basil, but when Milette shows up it is conclusive?
ReplyDeleteAnd for crying out loud, Milette worked on WTC dust before and his work is part of the official explanation, including lies to put the rescue workers to work without protection, knowing the dust was highly toxic.
JREF picked THIS guy after all the BS attacks against Steven Jones and Bentham. You really know how to pick them..
most likely the only guy willing to do such an experiment willing to overlook the ignition part of the study.
Do you want people to take you seriously - because if this is the best you can do it does not look like you are serious.
1. The "French dude", Frédéric Henry-Couannier, did not confirm Harrit's findings. Here is his presentation: http://www.darksideofgravity.com/marseille_gb.pdf
DeleteThis presentation has a page "Problems with the nanothermite hypothesis" (p.24), and on it, Henry-Couannier writes: "I could NOT confirm a reaction producing molten iron." (my upper-casing). So you are WRONG on the claim that Henry-Couannier confirmed Harrit.
2. If Mark Basile confirmed Harrit, as both Basile and Harrit claim, then ... well, Basile proves that less than 5% of the mass, and less than 2% of the energy of the red layer can come from thermite. See my blog post http://oystein-debate.blogspot.de/2012/03/how-mark-basile-confirms-that-red-gray.html
3. Can you tell me one lie Millette published on WTC dust, and source that claim? I think you are wrong on that count. Not even Kate Jenkins, in her very lengthy accusation, mentiones Millette, other than presenting him as a respected and accomplishes scientist. Please make sure your slander isn't libel!
4. Why has no one in the TM been able to pick an independent lab?
5. Millette is not overlooking the "ignition part" of the study. I hear he is currently looking into the microspheres issue and will publish a second preliminary report, hopefully including something on microspheres production, later in april. However, Millette understands that the DSC test as such are worthless. I will publish a blog post on that, too, but it will take me at least a week from now, because there is so much wrong with what Farrer did with the DSC, it is really some work to structure that.
6. Yes, what we do is very good and has already received respect and serious attention from some of the smarter members of the truth movement, who start to question why Harrit, Jones and Farrer are holding back TEM and FTIR data, why they and Ryan never release any dust to independent testers, why Gage is unwilling to pay for an independent test, and why basically nothing has happened in 3 years since the publication of the Bentham paper.
You come here basically with nothing but hand waving and slander. Is that the best YOU can do?
ADDENDUM:
Delete7. For the value of the accusations of fraud against Dr. James Millette, please red this: http://forums.randi.org/showthread.php?t=212725&page=86
Posts posts #3435-3537 by Chris Mohr.
'If you think the "official story" is wrong, and you can prove it, do it! What is your'
ReplyDeleteA bit off topic but I got to try this..
Official story= nobody knew nothing
Reality:
1. the gov knew about attacks but did nothing
- warnings from own agents and foreign agencies
knowledge from whistleblowers
2. the gov gave the hijackers visas to the US through the state dep to learn how to fly and plan mission - helped to train them
knowledge from whistleblowers
3. the government prevented FBI agents and others from investigating and stopping them
knowledge from whistleblowers
4. Osama worked for the US on 9/11
knowledge from whistleblower
5. Members of the 9/11 Commission have admitted that the official story is based on a lie..that is almost entirely untrue from start to finish because the gov lied to them at every stage of the investigation
6. People have come forward, including military brass, that believe it was an inside job. One recently stated that he knows who some of the traders are and is prepared to testify under oath in court
7.NIST was obliged by regulations to check for thermite since there was molten metal and bomb witnesses, but it did not look because it claimed there was not molten metal and no witnesses....this has proven to be a lie because they hid recordings of firefighters and others on 9/11 who said in no uncertain terms that the buildings were blown down...and there was molten metal afterwards. NIST lied and is stil lying.
8. witnesses, the red chips, molten spheres that were almost 6% of the dust, and signature air pollution all point to nanothermite made by US army labs.
9. It has already been proven that the anthrax was made by us army labs...
10. insider trading leading to Bush family, PNAC and lots of interesting bastards...completely white-washed by the commission
Are you blind? Are you KIDDING?
Ever heard of conlicts of interest?
The Commission was headed by one of the authors of the PNAC doctrine that dreamt about an attack to justify more wars and military spending...
The PNAC factions of the government is nr on the list of the usual suspects when it comes to inside job...so you have PNAC making sure no-one investigates PNAC
NIST investigates collapse of Towers but refuses to test for nanothermites - which NIST helped develop...
its investigation also taken over by a hoard of scientists from the very company that supplied falsified data to implicate Saddam to certain weapons that were never found...
GET A CLUE BUDDY - THERE NEEDS TO BE A NEW INVESTIGATION BEFORE OBAMA GROWS A FULL MUSTACHE!
ARE YOU NOT SEEING THAT THE US IS FALLING APART BECAUSE OF THIS 9/11 BS?
A bit off-topic? Ahem...
DeleteMy blog focusses on some technical aspects argued by CD and thermite proponents. I show that they are wrong, very wrong.
You can blind yourself to the fact that the leading proponents of these theories are very wrong, but some day you ought to wake to the fact that you have been lied to for years, and are still being lied to, by these leading truthers. You need to come to grips with this and ask why you should believe their political pronouncements when they lie to you about cold scientific facts.
'A bit off-topic? Ahem...My blog focusses on some technical aspects argued by CD and thermite proponents. I show that they are wrong, very wrong.'Oystein
ReplyDeleteYou make me laugh, the heading for your blog is:
'Debates between me and anybody who doubts the common narrative of the events of 9/11. If you think the "official story" is wrong, and you can prove it, do it!'
You cant have the debate you challenge others for, because you dont know shit about 9/11. Even the head of the 9/11 Commission has admitted that the 'common narrative' is a lie.
You have backed down from your challenge...
get a clue buddy...or stick to the kids at the JREF forum...they won't be bothered by your BS.
Government whistleblowers have confirmed that the gov got the hijackers in the US for training and prevented the FBI and others from stopping them....can you spell 'inside job' buddy?
- And you keep lokking for paint you fool!
Get a clue buddy
Anonymous,
Deleteyou quote-mine the header of my blog. You did quote "Debates between me and anybody who doubts the common narrative of the events of 9/11. If you think the "official story" is wrong, and you can prove it, do it!". You left out "What is your one (1) single most convincing argument? Your strongest evidence? Your most damning fact? State it as precisely as you can, and convince me!"
Please note the focus on the word "one". You presented me with a list of 10 reasons. And all ten are off-topic to the one technical aspect in the post were currently commenting on, namely "There are much more different kinds of red-gray chips than one". So if you believe that your "your one (1) single most convincing argument" against the official story is "there is only one kind of red-gray chios in the dust (and it is thermitic)", then you are indeed addressing the heading of this blog AND the current post. If not then not.
I'll tell you what: Tell me which is your one (1) single most convincing argument vs. official story, sign in to Blogspot, present your argument as a new post, and we'll debate your one (1) single most convincing argument there (and nothing else! No moving goal posts! No Gish Gallop!). Would that be acceptable? You set the topic, I try to prove you are wrong.
'Millette is not overlooking the "ignition part" of the study. I hear he is currently looking into the microspheres issue and will publish a second preliminary report, hopefully including something on microspheres production, later in april. However, Millette understands that the DSC test as such are worthless'
ReplyDeleteThese sort of statements make you look retarded, or I guess 'troll' is the accepted word.
Refusing to do the ignition test is overlooking the ignition part by definition, no matter how hard you look at the spheres that are left behind.
You get away with this sort of BS at JREF, but like I said, just about no-one else will take you seriously.
Arguing with people like you really IS a waste of time so let these be my last words...
Good Bye!
1. DSC is not an ignition test. Many DSC tests are totally without ignition. Can you explain to me what a DSC does?
ReplyDelete2. I understand Millette WILL ignite chips - he just won't do it with a DSC. Why would a DSC be necessary? Can you explain which addditional insight a DSC gives over other methods of ignition?
3. You are really running away from the debate before you have even argued your case? As far as I can see, so far you have merely stated your position, but presented no facts and no logic to support that position. Still, I take you serious.
Hi, Oystein,
ReplyDeletethanks for staying polite and factual even during discussion with this furious Anonym:o) But it is really not necessary to "take him serious" anymore, since he is not prepared to any meaningful debate, his posts are full of silly offenses and he is basically not able to understand anything what you have written.
Even "our" truthers in JREF are generally smarter and better informed.
It is perhaps a pity that you have not cought the attention of some educated truther so far. Anyway, thanks for your effort:o)
Ivan
Ivan,
DeleteI do this for the benefit of other readers, to demonstrate that the questions and objections raised by this anonymous commenter can be answered and / or have already been dealt with. I elected to ignore the offenses and address the technical content. It's easy.
Imagine you had the north tower intact as it was on 9/10/01. Say you could remove 5 levels 91 thru 95. That would leave 15 stories in the air without support and a 60 foot gap beneath.
ReplyDeleteIt would take 1.9 seconds for the 15 stories to fall 60 feet and they would impact at 42 mph or 62 ft/sec.
The 90 stories below would be about 1080 ft tall so if the 15 stories could maintain a constant velocity while crushing them the collapse should take 17.4 seconds plus the 1.9 seconds or 19.3 seconds. But Dr. Sunder of the NIST said the north tower came down in 11 seconds.
But completely eliminating 5 stories is more damage than the airliner and fires could do. So how did the towers come down so fast? And why don't we have accurate data on the distributions of steel and concrete down the towers after TEN YEARS?
psikeyhackr, do you wish for me to debate this as your contribution to my blog per the "What is your one (1) single most convincing argument?"? Then let's not do that here. This is the comments section to a post on red-gray chips, so you are off-topic.
DeleteIf you indicate you want to convince me of this one (1) argument, please indicate briefly with your next post, and I will leave you an email-address so we can discuss in private how to set up the debate.
Of course I am aware of several sound debunkings of your theory; you have a long history of fail, and perhaps I will simply link to some such debunkings. I am not sure I am actually going to waste time on this nonsense.
Oystein,
ReplyDeleteAnother well documented article.
'swelled to 5 x their size' in Methyl Ethyl Ketone - Ignition temp of MEK? 404 C. Autoignition of MEK 404 C - http://www.ppci.com.ph/msds2k10/08_mek.pdf
Jones found lying multiple times. Jones lies about Tritium. http://rense.com/general85/911.htm Covert ops hiding the nuke facts.
Related Articles
US Government's Usage of Atomic Bombs http://www.serendipity.li/wot/ed_ward/use_of_abombs.htm
Update: The US Government's Usage of Atomic Bombs – Domestic – WTC http://www.usavsus.info/WTC-MoreEvidence.htm
Update:Proven 9-11 Nukes = US Government Involvement http://edwardmd.wordpress.com/2010/09/06/proven-9-11-nukes-us-government-involvement/ Updated Broken Links
'9-11 Fake Video' Stars – The JSTAR Clones http://www.rense.com/general92/911fk.htm
Why Covert Ops Must Have `Fake' Video
No Windows on Flight 175 http://www.youtube.com/watch?v=jRC4lCQuBmc&feature=related
There was Nukes in Bali http://web.archive.org/web/20050129050637/http://homepage.ntlworld.com/steveseymour/nuke/bali_micro_nuke.htm , Nukes in London http://web.archive.org/web/20030409042415/http://geocities.com/vialls/nukes1.html , Nukes in Indonesia http://web.archive.org/web/20050213043930/http://www.vialls.com/myahudi/embassynuke.html , etc, et al? Where was Nukes in NYC?
Ed Ward, MD
Ed Ward MD's Weblog of Tyranny http://edwardmd.wordpress.com/ http://www.facebook.com/EdWardMD3 http://groups.yahoo.com/group/EdWard-MD/
Ed, you are plain nuts.
DeleteSure, Jones is lying. At least he is very wrong. Jones slams Fetzer, Fetzer slams Jones. Since Jones is wrong, is Fetzer right? No, of course not, Fetzer is even more dramatically wrong. You slam Jones, Jones slams nukers. Are you right because Jones is wrong? No, of course not. You are the wrongest of them all.